
Yet oxidation values can vary depending on which non-metal elements they are combined with, as in polyatomic ions and molecu-lar compounds. Non-metal elements have a common or fixed charge/ oxidation when compounded with metals. When atoms (typically metals) lose one or more electrons the become positively charged cations. For ions in group VA, VI, and VIIA, the magnitude of charge is calculated by subtract-ing the group number from 8. These different types of ion forming metals will be discussed more in the next section on naming compounds ( section 2.7.3). Monatomic or monoatomic elements are elements that are stable as single atoms. Third, there are two types of metals, those that have only one charge state (like the nonmetals), and those that have multiple charge states. Second, the nonmetals have only one charge state, which represents the number of electrons that need to be added for them to have the same number as a noble gas. First, metals tend to from cations and nonmetals tend to form anions, while the noble gasses do not tend to form ions. There are several things you can note from this table though. The charge of an electron is considered to be negative by convention and this charge is equal and opposite to the charge of a proton. If the ion of element X has a mass number of 230 and 86 electrons, what is the identity of the element, and how many neutrons does it have Which atom is most likely to form a 2+ ion (a) Zn. An ion ( / a.n, - n /) 1 is an atom or molecule with a net electrical charge. An elements most stable ion forms an ionic compound with bromine, having the formula XBr2. 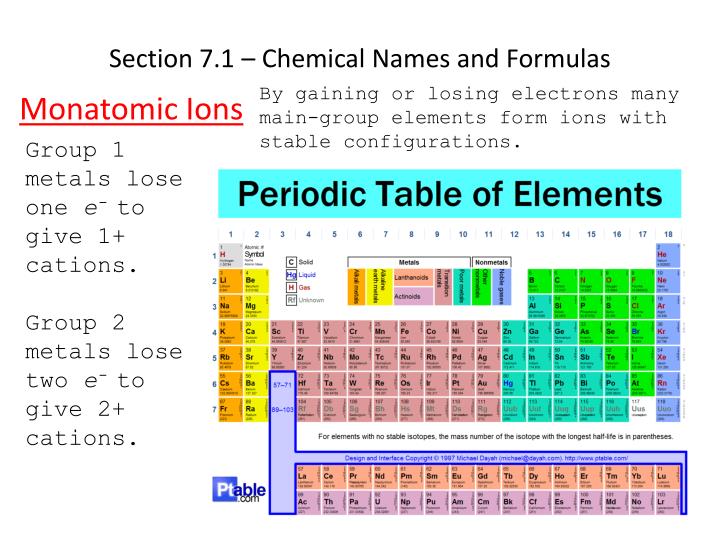
sodium selenium barium rubidium nitrogen aluminum 8. Forming an ionic bond, Li and F become Li + and F ions. Predict the charge on the most common monatomic ion formed by each element. 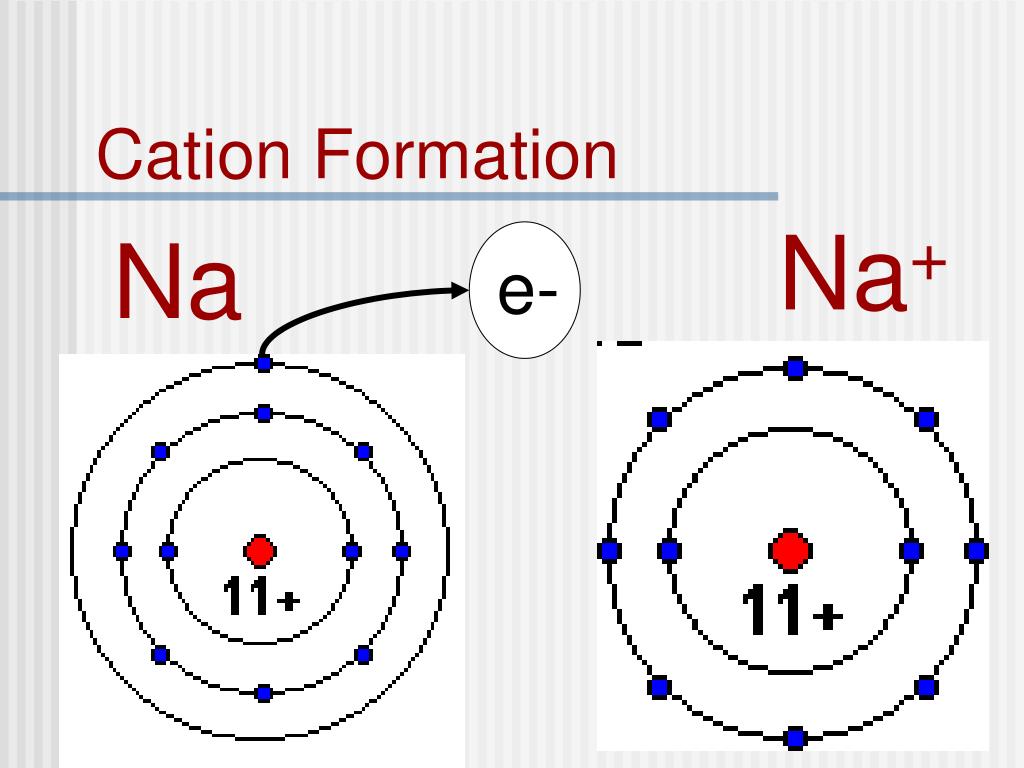
All atoms try to achieve that by bonding with other atoms. Wiki User 00:39:42 Study now See answer (1) Best Answer Copy. \) should not be considered to be a comprehensive Figure and this topic will be brought up again in the next section. Electron transfer between lithium (Li) and fluorine (F). Noble gas configuration is the most stable electron configuration. Atomic Ionic radius, r ion, is the radius of a monatomic ion in an ionic.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
